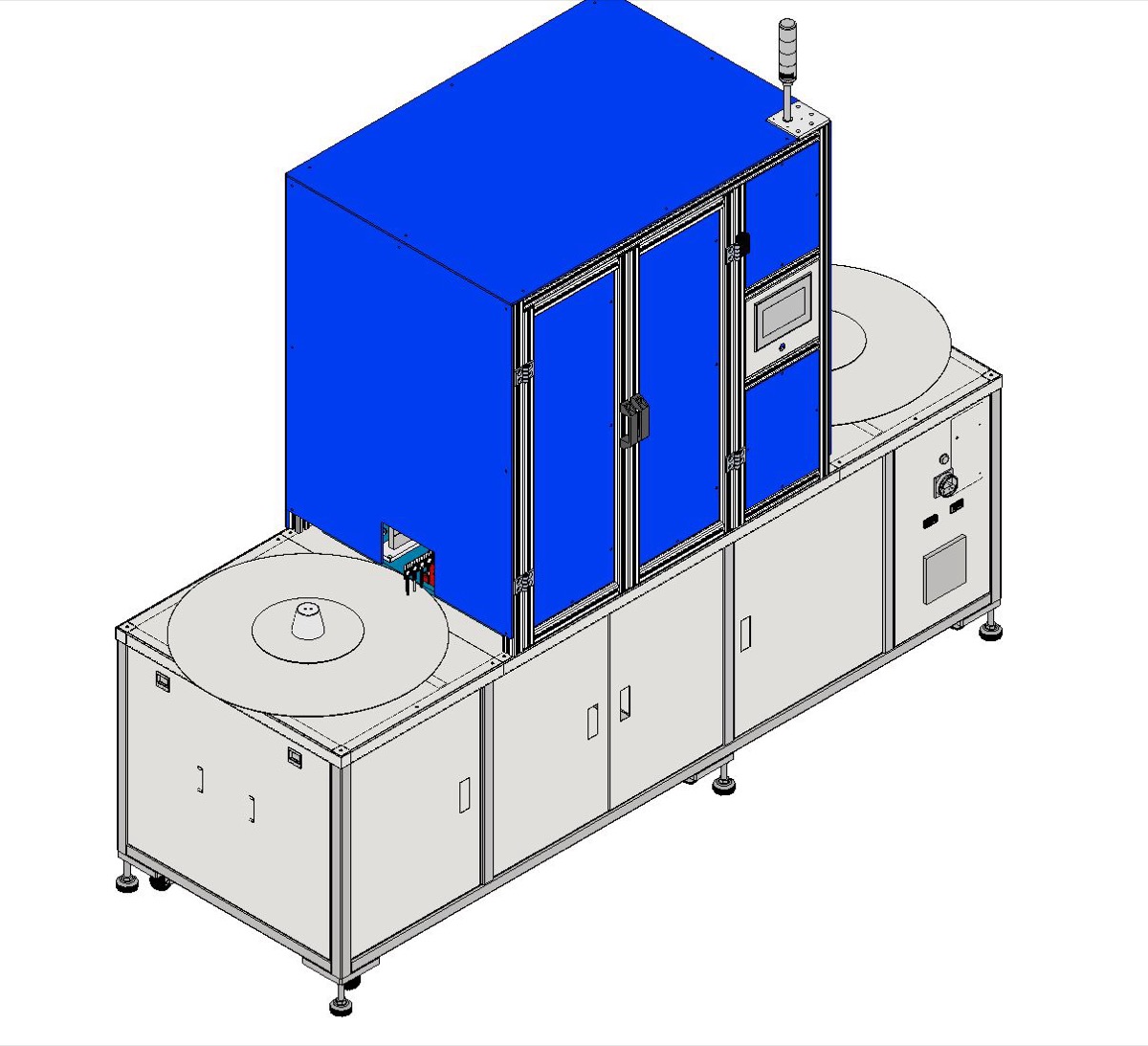
Motionwell designs and builds pharmaceutical packaging machinery with GMP-grade stainless steel construction (SUS304/316), precision volumetric and gravimetric dosing, and integrated inspection at every critical point. Our delivered systems include triple-head simultaneous filling machines running at 20-60 units per minute, automated vial capping and uncapping stations (P23005), and dispensing units with cartridge and pump assemblies (P25026).
We build for what matters in pharmaceutical packaging: fill accuracy within ±1%, clean material paths with no dead zones, and batch reporting that supports your compliance documentation.
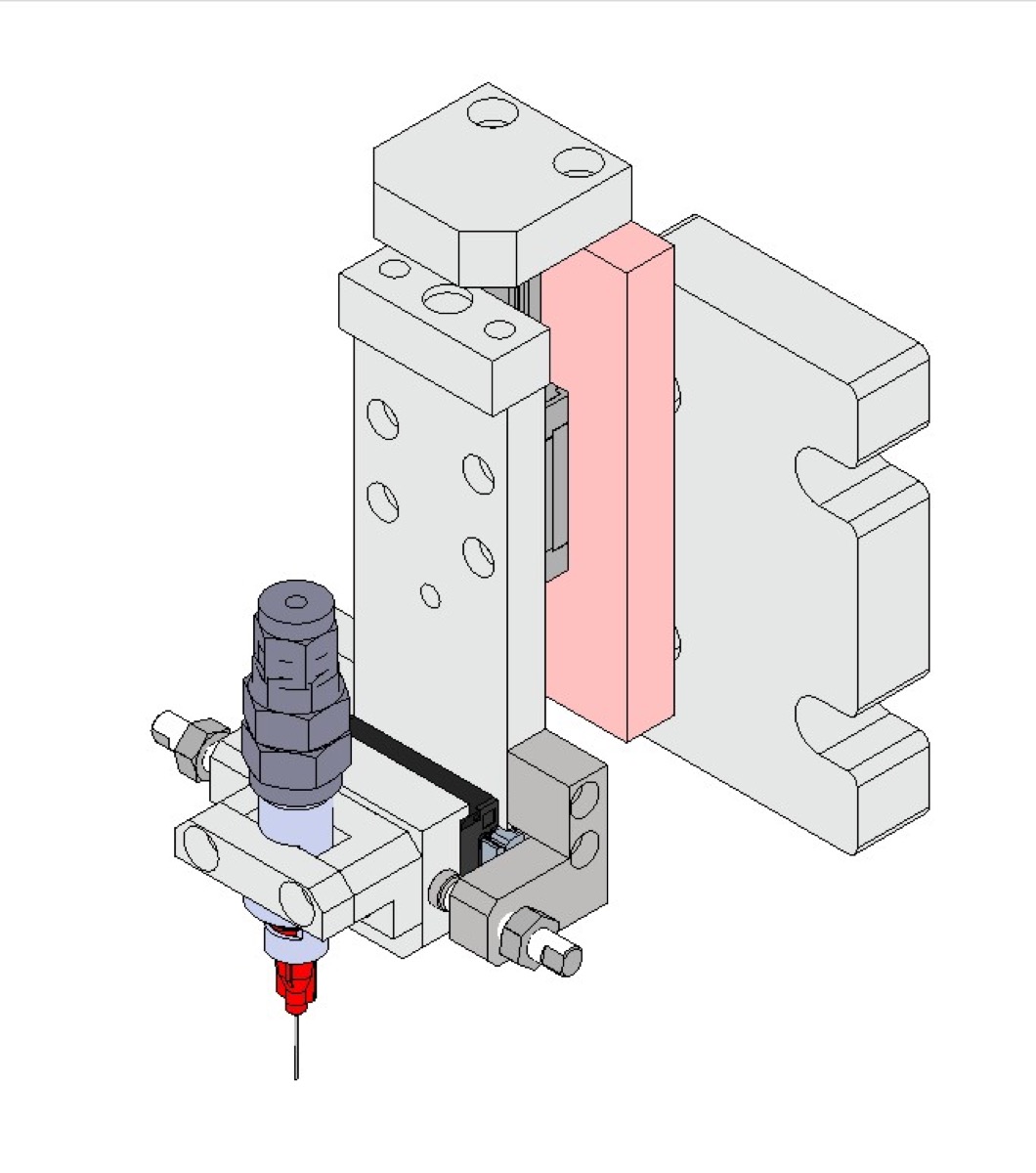
- Triple-head simultaneous filling with volumetric and gravimetric dosing (±1% accuracy, 20-60 pcs/min) for oral liquids, eye drops, and reagent vials
- Automated vial capping and uncapping stations (P23005) with torque control and cap presence verification
- Serialization and coding with TIJ/laser printing, OCR/OCV verification, and red strobe inspection for track-and-trace compliance
- Continuous dual-tray feed design for non-stop filling operation with automatic tray changeover
- GMP-aligned SUS304/316 stainless steel construction with aseptic filling capability and no-dead-zone fluid paths
- Verification at every stage: fill level, seal integrity, label accuracy, and code readability
- Maintenance-friendly design with tool-free access panels, clean-in-place support, and safe changeover procedures
- Clear fault diagnosis with HMI alarm codes, fast recovery sequences, and batch reporting (counts, rejects, reason codes)
- Servo-driven dispensing with level sensors, recipe-based parameter control, and automatic tare correction
- Conveyor indexing with heat seal, crimp seal, and ultrasonic seal integration
- Vision systems for print and label verification with programmable reject gates and pneumatic segregation
- PLC recipe management, alarm handling, event logging, and 21 CFR Part 11 batch record support
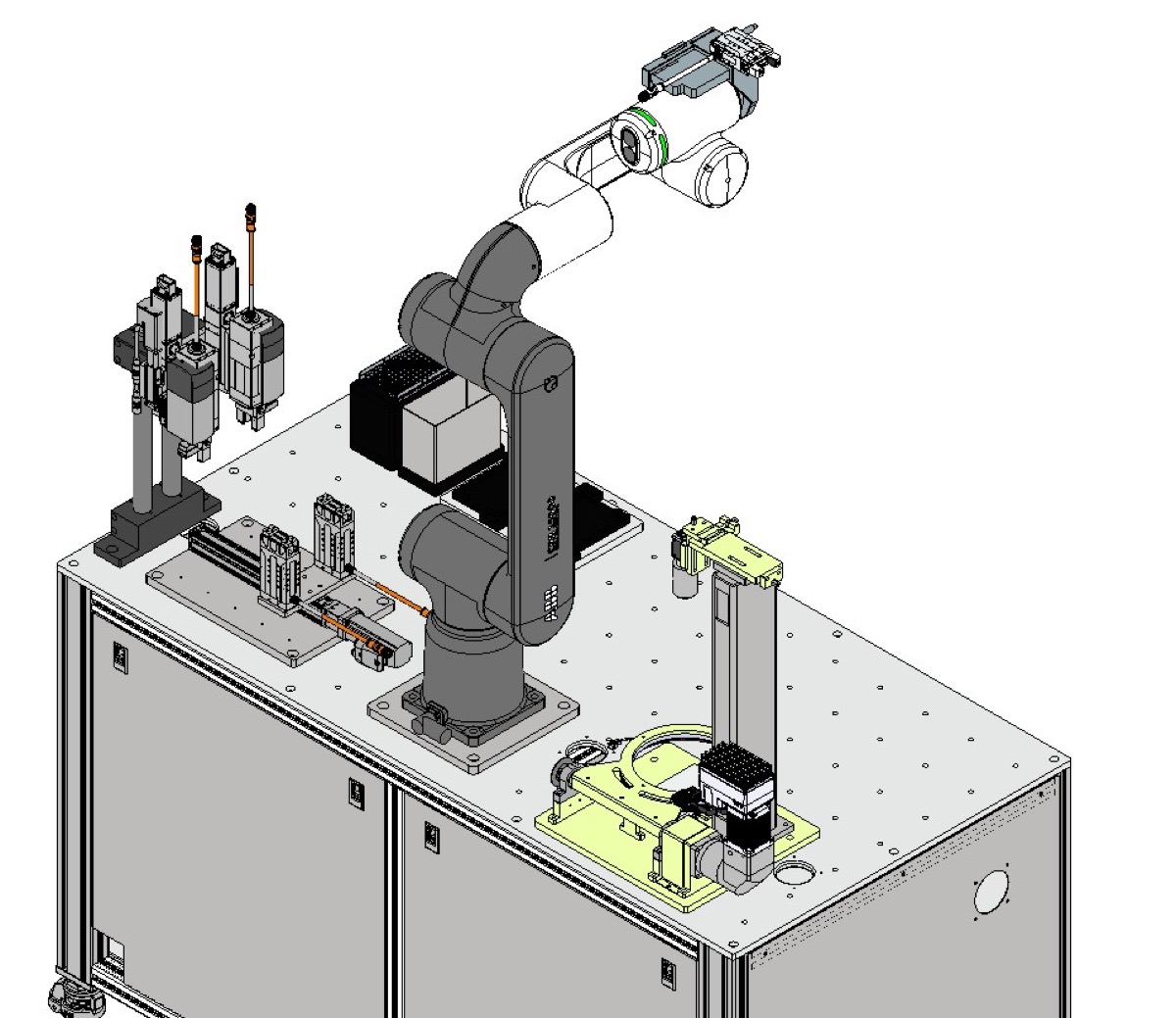
Featured: Triple-Head Precision Filling & Sealing Machine
Motionwell designed and built a precision filling and sealing machine for pharmaceutical oral liquids with triple-head simultaneous dosing. The machine features SUS316L stainless steel contact surfaces, continuous dual-tray feed for non-stop operation, and integrated heat sealing with reject sorting.
- Output: 20-60 units/min with triple-head simultaneous fill
- Accuracy: ±1% volumetric and gravimetric dosing
- Construction: SUS304/316L frame and contact surfaces, GMP-grade finish
- Features: Recipe-based HMI, batch reporting, automatic reject segregation