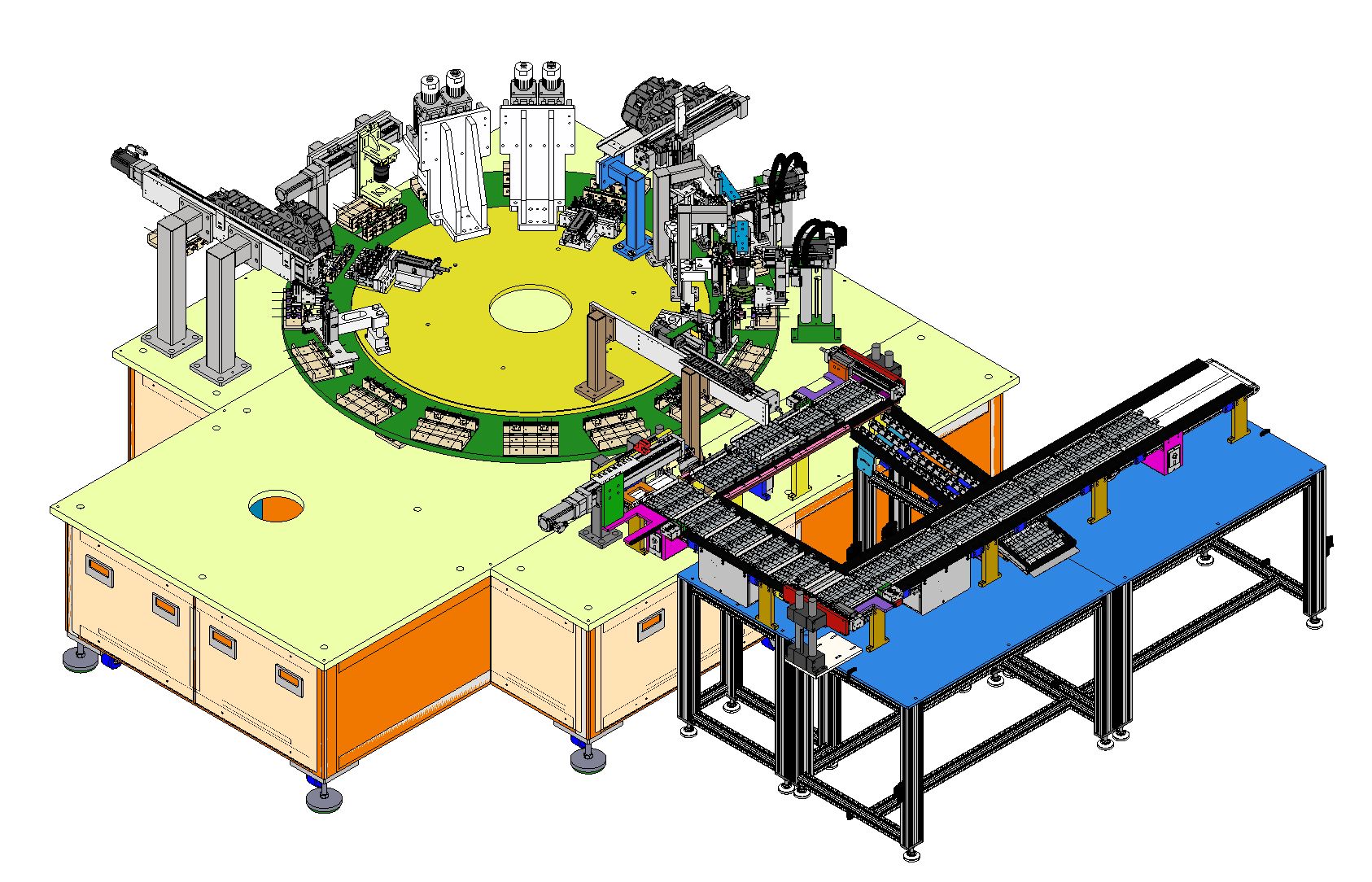
Project Overview
Motionwell Automation designed and built GMP-grade filling and sealing machines (Projects P23005, P25026) for pharmaceutical manufacturers in Singapore. The machines handle liquid product filling with servo-driven volumetric accuracy and automated sealing with rotary capping heads.
GMP-Compliant Design
Material Specification
The machine frame and all product-contact components are fabricated from SUS316L stainless steel (1.4404 grade) with electropolished surfaces to Ra 0.4um or better. Non-contact structural members use SUS304 to manage material cost while maintaining corrosion resistance in washdown environments:
- All welds ground flush and passivated per ASME BPE standards for pharmaceutical equipment
- Garlock Gylon 3545 PTFE gaskets and Parker E-series EPDM O-rings at all product-contact sealing surfaces, both FDA 21 CFR 177.2600 compliant
- Clean-in-place (CIP) compatible design with spray ball ports at all dead legs and drain points sloped at minimum 3 degrees
Filling System
The filling stations use Festo EMMS-AS servo motors driving ceramic-lined piston pumps for volumetric dispensing. Each fill head operates independently, allowing per-nozzle volume calibration:
- Filling accuracy within +/-0.5% of target volume across the 5ml to 500ml range
- 8-head rotary filling configuration achieving 120 bottles per minute at 100ml fill volume
- Keyence FD-Q series flow sensors provide real-time fill volume verification as a secondary check
- No-bottle-no-fill detection using Omron E3Z retroreflective sensors at each fill position
- Anti-drip nozzles with pneumatic shut-off valves and suck-back function to prevent product stringing
Sealing System
The sealing section handles both ROPP (roll-on pilfer-proof) aluminum caps and screw-on plastic closures via quick-change capping chuck assemblies:
- Rotary capping heads driven by Mitsubishi HG-KR servo motors with torque feedback
- Programmable torque range from 0.5Nm to 5.0Nm with real-time torque curve logging
- Cap presence verified by Keyence IL-600 laser displacement sensor measuring cap height
- Automated reject station with pneumatic pusher directing improperly sealed containers to a quarantine bin
Serialization Integration
The line integrates with Cognex DataMan 370 fixed-mount readers and Domino Gx150i thermal inkjet printers for track-and-trace serialization compliant with Singapore HSA and EU FMD requirements:
- 2D DataMatrix code printing and verification at up to 150 units per minute
- Aggregation data logged to a Level 3 serialization server for parent-child relationship tracking
- Reject mechanism with Festo DSBC pneumatic cylinders for unreadable or duplicate codes
GMP Validation
Motionwell provides full IQ/OQ/PQ (Installation Qualification, Operational Qualification, Performance Qualification) documentation packages as part of the machine delivery. Factory acceptance testing (FAT) is conducted at Motionwell’s facility in Singapore prior to shipment, followed by site acceptance testing (SAT) at the customer’s cleanroom. All validation protocols follow ISPE Baseline Guide Vol. 5 for commissioning and qualification.
Related
This project showcases Motionwell’s custom machine design and machine vision inspection expertise for the pharmaceutical & packaging industry.