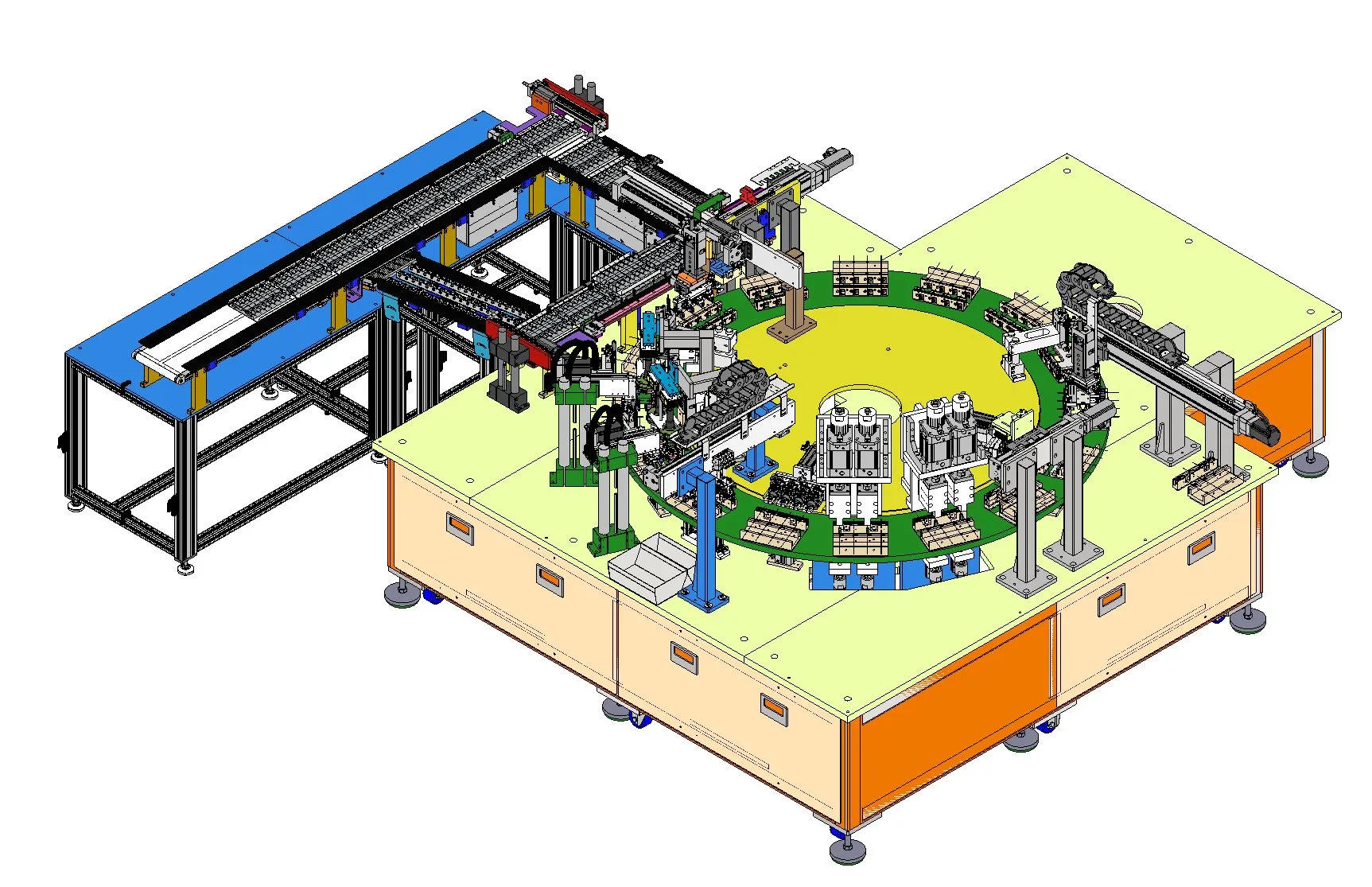
Project Overview
Motionwell Automation designed and built a 12-station rotary indexing assembly machine (Project P22068) for a global medical device manufacturer. The machine assembles safety syringes and blood collection tubes at a cycle time of 15 seconds per part.
The project required integration of multiple automation technologies: servo-driven rotary indexing, vibratory bowl feeding, Epson SCARA robotic pick-and-place, and medical-grade vision inspection. All components were designed for ISO Class 7/8 cleanroom operation.
Machine Design
12-Station Rotary Indexing Table
The core of the machine is a precision rotary indexing table with 12 work stations. Each station performs a specific assembly or inspection operation:
- Component loading via vibratory bowl feeders
- Servo-driven assembly with controlled force profiles
- Multi-point vision inspection (dimensional, presence, orientation)
- OK/NG sorting with automated reject handling
- Epson SCARA robot for final pick-and-place
Vision Inspection System
The vision inspection system meets stringent medical device quality standards with:
- Keyence vision cameras at multiple stations
- Contour recognition for dimensional verification
- Presence and orientation detection
- Automated OK/NG sorting with reject bins
- Full traceability of inspection results
SCARA Robot Integration
An Epson SCARA robot handles high-speed pick-and-place operations at the output station, sorting assembled products by inspection result and loading into packaging trays.
Technical Specifications
| Parameter | Specification |
|---|---|
| Stations | 12-station rotary index |
| Cycle Time | 15 seconds/part |
| Robot | Epson SCARA |
| Vision | Keyence cameras, medical-grade |
| Cleanroom | ISO Class 7/8 compatible |
| Construction | Stainless steel, poka-yoke |
Technical Details
Servo-Driven Indexing Mechanism
The rotary indexing table uses a servo-driven cam indexer rather than a pneumatic or Geneva mechanism. The servo motor provides programmable motion profiles with controlled acceleration and deceleration, eliminating the mechanical shock of fixed-dwell indexing. Indexing repeatability is +/-0.05mm at each station position, verified during commissioning with a laser tracker measurement over 1,000 consecutive index cycles.
The servo drive enables variable dwell time at each station, allowing longer-duration operations (such as press-fit or vision inspection) without penalizing the cycle time of faster stations. Motion profiles are stored as recipes in the PLC and can be adjusted without mechanical modification.
Jig and Fixture Materials
All workholding jigs on the rotary table are machined from nylon (PA66) or POM (polyoxymethylene/acetal) engineering plastics. These materials were selected for two reasons:
- Electrical insulation: Nylon and POM prevent static discharge that could damage sensitive medical device sub-components during assembly. Surface resistivity exceeds 10^12 ohm, meeting ESD-safe handling requirements for the cleanroom environment.
- Wear resistance: POM provides excellent dimensional stability and low friction coefficient (0.2-0.35 against steel), ensuring consistent part location over hundreds of thousands of cycles without generating particulate contamination that would compromise cleanroom air quality.
Jig contact surfaces are precision-machined to +/-0.02mm tolerance with polished finishes to prevent marking or scratching product surfaces.
Pneumatic Press-Fit with Force Monitoring
The assembly stations that perform component insertion use pneumatic press-fit actuators with integrated force monitoring. Each press cylinder is equipped with a strain-gauge load cell that measures insertion force in real-time throughout the press stroke. The PLC records the complete force-displacement curve for every assembly cycle.
The system implements closed-loop feedback control: if the measured insertion force exceeds or falls below the programmed tolerance window at any point during the stroke, the press immediately halts and the part is flagged for rejection. This catches conditions such as misaligned components (force too high), missing components (force too low), or damaged parts (irregular force profile). Force data is logged per serial number for full traceability.
Vision System Lighting
The Keyence vision inspection stations use dome lighting (diffuse hemispherical illumination) rather than ring lights or bar lights. Dome lighting creates uniform, shadow-free illumination across the entire inspection field of view. This is critical for medical device inspection where:
- Component edges must be measured without shadow-induced dimensional errors
- Surface defects (scratches, contamination, flash) must be detected uniformly regardless of their angular orientation on the part
- Translucent or reflective medical-grade materials (polycarbonate, stainless steel) would create specular highlights under directional lighting that interfere with contour recognition algorithms
The dome lights use white LED arrays with diffuser panels, providing consistent color temperature (6500K) for repeatable color-based inspection criteria.
Validation Documentation
Motionwell delivers the machine with a complete IQ/OQ/PQ (Installation Qualification, Operational Qualification, Performance Qualification) validation documentation package. The IQ verifies that the machine is installed per the design specification, including utility connections, safety system wiring, and software version control. The OQ confirms that each station operates within specified parameters under worst-case conditions. The PQ demonstrates sustained production capability over a minimum of three consecutive production runs.
All validation protocols are prepared in compliance with GAMP 5 guidelines for automated systems in regulated manufacturing. The documentation package includes test protocols, executed test records, deviation reports, and a final validation summary report suitable for regulatory audit.
Related
This project demonstrates Motionwell’s custom machine design and machine vision inspection expertise for the medical device industry.